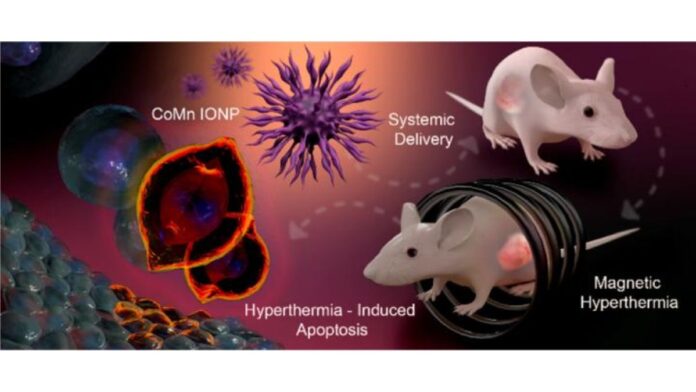
Researchers at Oregon State University have improved techniques that use magnetic nanoparticles to kill cancer cells with heat.
The improved magnetic nanoparticles – particles that measure close to one-billionth of a meter- created by these researchers generate heat in the cancer cells when activated using an alternating magnetic field.
This form of cancer treatment is a promising and developing method of cancer therapy known as magnetic hyperthermia. It is based on the observation that when heated to temperatures ranging from 42 to 46°C, cells undergo programmed cell death or apoptosis. In recent years, there have been successful preliminary clinical trials using magnetic hyperthermia to treat patients with glioblastoma and prostate cancer. Despite these successes, there are multiple roadblocks to be overcome before magnetic hyperthermia is used as a mainstream cancer therapy.
One of the primary roadblocks is the effective targeting of the magnetic nanoparticles in cancer cells. Currently, these nanoparticles are delivered to localized and relatively accessible tumors by directly injecting the nanoparticles into the tumor. However, this method is not useful in targeting tumors that are deep inside the body.
Intravenous (IV) administration is a viable method to deliver these nanoparticles to inaccessible and poorly defined tumors including primary tumors and small metastatic growths. A challenge with the IV method of delivery is the relatively low levels of accumulation of the nanoparticles in the tumor cells.
Additionally, the commonly used iron oxide nanoparticles (IONP) do not heat up sufficiently to effectively kill the tumor cells. To overcome these challenges researchers at Oregon State University led by Dr. Oleh Taratula modified the composition and shape of the iron oxide nanoparticles, which resulted in improved heating efficiency.
The hexagon-shaped cobalt and manganese ‘doped’ iron oxide nanoparticles (CoMn-IONP), upon activation by use of an alternating magnetic field, were shown to effectively kill cancer cells in cell culture as well as mouse models. These findings were published in ACS Nano.
The nanoparticles reduce tumor volume in mice
Experiments using cancer cells grown in a petri-dish showed that the modified CoMn-IONP increased the cellular temperature by 17°C, raising the temperature to 46°C, within eight minutes. In contrast, unmodified iron oxide nanoparticles only increased the temperature by 3°C. Next, the researchers injected the nanoparticles into mice that had ovarian tumors grafted under their skin.
The nanoparticles accumulated primarily in the tumor cells within five hours of injection. These nanoparticles were activated by the use of an alternating magnetic field for 30 minutes and this was repeated once a week for four weeks. About thirty-five days after treatment, the tumor volume in mice treated with CoMn-IONP was five times smaller than in mice treated with unmodified IONP.
The nanoparticles do not damage healthy cells
The researchers also noted that the nanoparticles were capable of killing cancer cells only upon activation and their mere presence is not toxic to the mice. The nanoparticles did not adversely affect the non-cancerous, normal cells.
The nanoparticles were also shown to not affect the genetic information in cells, thus reducing the fear of introducing unwanted mutations in cells.
The promising results in this study bode well for the use of magnetic hyperthermia in treating different types of tumors, including tumors inaccessible by surgery or metastatic growths. As Dr. Olena Taratula, one of the lead authors of the study notes in the press release, “Our new nano platform is a milestone for treating difficult-to-access tumors with magnetic hyperthermia. This is a proof of concept, and the nanoclusters could potentially be optimized for even greater heating efficiency.”
However, there are several factors that still need to be addressed in order to improve the safety of the magnetic hyperthermia treatment method.
For instance, upon activation of the nanoparticles heating of the healthy cells that are adjacent to the tumor cells can occur. Currently, there is no consensus on the strength of the activating alternating magnetic field that can be safely tolerated. Further clinical studies will need to be carried out to develop the treatment standards as well as optimization of systems that deliver the alternating magnetic field to only the tumor region and reduce heating in the normal tissue.
As explained by Dr. Olena Taratula, “To advance this technology, future studies need to use orthotopic animal models – models where deep-seated tumors are studied in the location they would actually occur in the body.” She added, “In addition, to minimize the heating of healthy tissue, current AMF systems need to be optimized, or new ones developed.”
Written by Bhavana Achary, PhD
References:
Albarqi HA, Wong LH, Schumann C, Sabei FY, Korzun T, Li X, Hansen MN, Dhagat P, Moses AS, Taratula O, Taratula O. Biocompatible Nanoclusters with High Heating Efficiency for Systemically Delivered Magnetic Hyperthermia. ACS Nano. 2019 Jun 25;13(6):6383-6395
Chang David, Lim May, Goos Jeroen A. C. M., Qiao Ruirui, Ng Yun Yee, Mansfeld Friederike M., Jackson Michael, Davis Thomas P., Kavallaris Maria. Biologically Targeted Magnetic Hyperthermia: Potential and Limitations.Frontiers in Pharmacology. 2018. Volume 9, p831.
Press release: Researchers reach milestone in use of nanoparticles to kill cancer with heat. Available at: https://www.eurekalert.org/multimedia/pub/204856.php?from=434149
Photo credit: Tetiana Korzun