A recent study investigated the association between the risk of potential side effects of donepezil, a popular drug used to treat symptoms of dementia, and hospital admissions.
Dementia is prevalent in the aging population around the world with nearly 10 million new cases each year. The condition is described as a set of symptoms that impact a person’s cognitive abilities such as memory as well as communication and the ability to perform daily activities. Alzheimer’s disease is a brain disease that gets worse with time and accounts for the majority of the cases of dementia.
Donepezil, a cholinesterase inhibitor, is one of the most commonly prescribed medications to treat symptoms of Alzheimer’s disease and other dementias. Health Canada and regulatory agencies in other countries raised concerns about the risk of rhabdomyolysis as side effects of donepezil based on case reports from around the world. According to the agencies’ concerns, most cases dealing with the side effect occurred after either first taking the drug or after increasing the dose.
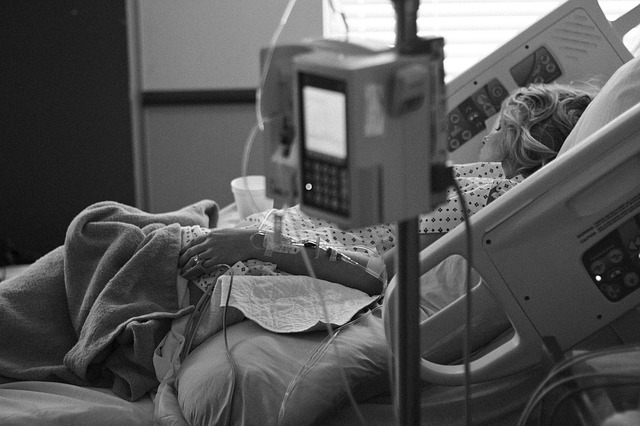
Rhabdomyolysis is a condition dealing with the breakdown of damaged skeletal muscle and can lead to kidney damage. The condition can be treated with success if caught early in its progression. The treatment option for most cases is intravenous therapy (IV), which involves delivering fluids to patients through their veins. In severe cases, patients may require acute dialysis to treat kidney damage.
In a study published in the Canadian Medical Association Journal, researchers aimed to determine whether the risk of hospital admissions with rhabdomyolysis was associated with donepezil in comparison to other commonly used cholinesterase inhibitors such as rivastigmine or galantamine. The study examined a cohort of patients aged 66 or older, residing in Ontario, Canada and who were issued newly prescribed donepezil, rivastigmine, or galantamine. The information on the patients was taken from various healthcare databases and included over 220,000 persons from 2002 to 2017. The patients were divided into two groups. Patients who were prescribed donepezil were placed in one group and the ones prescribed with rivastigmine or galantamine were put in another group.
The results of the study showed that the risk of hospitalizations with rhabdomyolysis was doubled with donepezil in comparison to rivastigmine or galantamine. The researchers found that most of the hospital admissions within 30 days of starting the medication did not report severe cases of rhabdomyolysis and none of the patients were found to be treated for kidney damage. The study reported that while the risk of hospitalizations associated with donepezil was small, it was statistically significant compared with rivastigmine or galantamine.
The study by having a large sample size of over 150,000 patients using donepezil had given the researchers the opportunity to investigate a potential side effect that might pose serious risks to the patient’s health. The authors of the study conclude, “The findings of this population-based cohort study support regulatory agency warnings about the risk of donepezil-induced rhabdomyolysis.” Furthermore, the risk of developing severe side effects of donepezil within 30 days of initiating the drug was low.
Written by Ranjani Sabarinathan, MSc
References:
Fleet, J.L., McArthur, E., Patel, A., Weir, M.A., Montero-Odasso, M., and Garg, A.X. (2019). Risk of rhabdomyolysis with donepezil compared with rivastigmine or galantamine: a population-based cohort study. CMAJ. doi: 10.1503/cmaj.190337
Commonly used drug for Alzheimer’s disease doubles risk of hospitalization. (2019, September 16). Retrieved from https://www.eurekalert.org/pub_releases/2019-09/ji-cud091019.php
Dementia and Alzheimer’s: What Are the Differences. https://www.healthline.com/health/alzheimers-disease/difference-dementia-alzheimers#dementia
Rhabdomyolysis: Causes, Symptoms, and Diagnosis. ttps://www.healthline.com/health/rhabdomyolysis
Image by Parentingupstream from Pixabay